
The thermal effects on cells depend mainly on two factors – temperature and time. Pre-clinical research done by, amongst others, our co-founder Prof. Karl-Göran Tranberg, has shown that moderate heating of cancerous tumor cells during a long time will kill the targeted tumor itself, and also activate the immune system to attack cancer cells throughout the body.
When cells of a cancerous tumor are moderately heated over time their membranes are cracked. The heated cell then opens and exposes the tumor-specific antigens that were contained within. The body’s immune system reacts to the exposed antigens by producing antibodies can recognize and “flag” these specific tumor antigens anywhere in the body. Thereby the immune system can recognize and attack remaining tumor cells, even those that are outside, or distant from, the treated area. This resulting phenomenon of immune stimulation is called the abscopal effect.
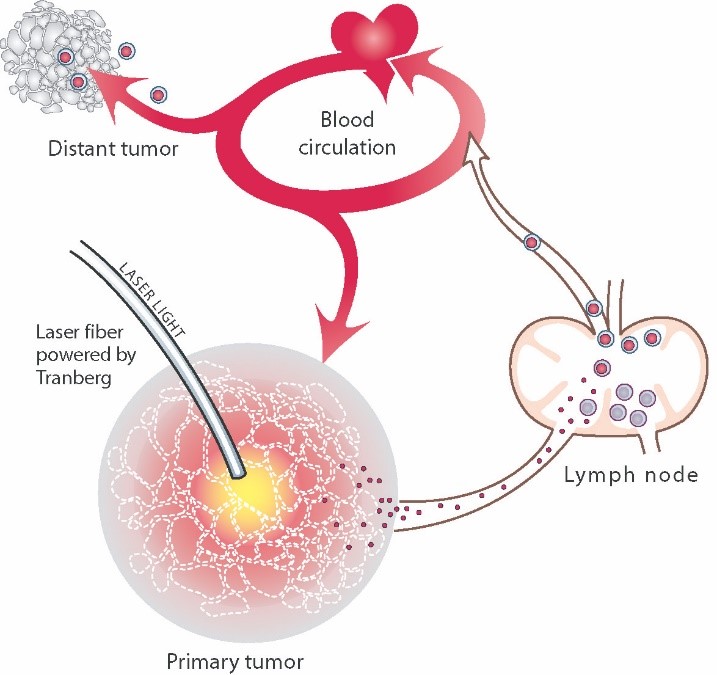
Even if the principle is simple, the procedure requires great precision and control. Thermal energy must be accurately guided into the tumor and precisely controlled to generate low, non-coagulating temperatures at the outer surface of the tumor. This causes the cell membranes in this area to open and antigens to be released, creating an immune response that potentially also attacks existing metastases in the body, the abscopal effect.
With the purpose of providing an abscopal effect to be used as a monotherapy or in combination with other cancer immunotherapies in curative treatment of metastasized (spread) cancer, CLS has developed its own specific immune stimulating ablation protocol named imILT®.

The imILT protocol is delivered through a minimally invasive image-guided procedure with the precision and accuracy required using the TRANBERG | Thermal Therapy Systems, with their built-in tissue temperature feedback and control features, integrated with an existing Ultrasound/MR scanner and instrument navigation and positioning system.
Note: The clinical effects when applying the imILT protocol are being investigated and the use of imILT is currently restricted to clinical investigations only.
VP Medical Affairs
kristian.waldeck@clinicallaser.com

Last week we joined the 2024 Congress of Neurological Surgeons (CNS) meeting in Houston where our partner ClearPoint Neuro, Inc. exhibited the ClearPoint Prism® Neuro Laser Therapy System, powered by CLS. In addition to good booth traffic and customer discussions, we also took part in the scientific program providing the latest updates on the use of Laser Interstitial Thermal Therapy (LITT) in neurosurgery.

Both abstracts provide the latest insights into the clinical use of focal laser ablation for prostate cancer, contributing to the ongoing development of minimally invasive cancer therapies.

On the 29th of August, CLS organized a training camp for our European distributors within urology and their Clinical Applications Specialists. During two intense days in Turin, Italy, the team learned a lot about the treatment of localized prostate cancer using the CLS TRANBERG® product portfolio, both in theory and in practice.
Welcome to Clinical Laserthermia Systems.
Please note that all our products and indications are not yet approved in all markets. Don’t hesitate to contact us for up-to-date market approvals in your area!
Please choose your country or region